Why Acertara Invested in MAUI Imaging — and What It Means for Hospitals
Why Acertara Invested in MAUI Imaging — and What It Means for Hospitals
Published – August 2025. Last updated – August 2025
Executive Summary
In July 2025, Acertara led MAUI Imaging’s $14 million funding round, strengthening its role in advancing innovative medical imaging. The MAUI K3900 holds FDA 510(k) clearance under K230511, with indications detailed in the FDA record. Acertara is managing pre-orders with an exclusive U.S. distributorship for hospitals outside of those MAUI serves directly. This role positions Acertara to coordinate U.S. market entry, oversee training and workflow integration, and support hospitals with acceptance testing, commissioning, and post-market surveillance. Together, these efforts align MAUI’s computed echo tomography technology with the operational and quality requirements of clinical engineering and healthcare technology management teams.
Table of Contents
Hospitals are continually balancing the need for speed, accuracy, and patient safety when choosing imaging tools. Ultrasound remains one of the most versatile modalities available, but its limitations become clear in scenarios where bone, surgical hardware, or air-filled cavities obstruct the acoustic pathway.
In trauma bays, neurosurgical suites, and interventional radiology (IR) labs, those barriers can delay diagnosis or force a modality switch — often to computed tomography (CT) or magnetic resonance imaging (MRI), which may require patient transport, additional resources, or exposure to ionizing radiation.
MAUI Imaging is taking a different approach. Its technology, cleared by the Food and Drug Administration (FDA) under K230511, is designed to visualize anatomy in ways conventional ultrasound cannot. This includes situations where bone or complex tissue interfaces limit standard acoustic penetration. For a closer look at how MAUI targets these needs, review the trauma, neurosurgery, and IR use cases.
For clinicians, the value is clear: improved intraoperative decision-making, faster point-of-care assessment, and the ability to guide procedures without leaving the clinical environment. For hospitals, it represents a tool that could potentially reduce patient transfers, free up high-cost imaging suites, and integrate into workflows already built around ultrasound portability.
Multiple OEM Certified
Industry Highest Successful Probe Repair Rate
Cloud-Based Documentation
13485:2016 Certified
Years in Business, Sonora + Acertara
Probes Repaired Since 1998
Understanding the Technology
MAUI’s K3900 platform is based on computed echo tomography (CET) — a patented approach that applies advanced reconstruction algorithms to acoustic signals. CET builds upon ultrasound’s non-ionizing safety profile while addressing some of its key limitations in complex anatomy.
Instead of relying solely on standard beam-forming, CET uses proprietary techniques to collect, process, and reconstruct echoes from multiple angles and depths. This can produce images that reveal structures otherwise hidden in shadow or distortion with 2D probes. Importantly, while CET is not intended to replace CT or MRI, its ability to operate at the point of care means faster access to critical information in time-sensitive scenarios.
MAUI has outlined the technical principles behind CET, but its clinical impact will be proven in real-world settings, particularly those that demand quick, accurate imaging where traditional tools fall short.

Acertara’s Strategic Role
Acertara’s decision to lead MAUI’s $14 million funding round in July 2025 extended beyond financial backing, aiming to ensure hospitals have the resources, training, and quality infrastructure to use CET effectively.
To help accelerate the adoption of this innovative technology, Acertara will:
- Coordinate pre-orders with hospital procurement teams to align with budget cycles, capital equipment planning, and grant timelines.
- Manage deployment logistics so MAUI systems arrive ready for site-specific setup and integration.
- Deliver training pathways for clinical users, from sonographers to surgeons, tailored to each department’s use cases.
- Integrate with QA workflows, using tools like ATLAS acceptance testing to benchmark performance during commissioning.
- Support probe service integration, leveraging Acertara’s industry-leading probe repair and performance testing capabilities.
- Feed post-market data back to MAUI for continuous product improvement, including insights from field service engineers, biomedical teams, and clinical users.
By combining MAUI’s innovation with Acertara’s quality assurance expertise, hospitals gain not just a new device but a partner who understands the operational realities of imaging technology adoption.
Regulatory Clarity: MAUI K3900
FDA-Cleared Indications for K3900 (K230511)
- For use in adult and pediatric populations
- Intended for transcranial imaging of cerebral structures through the temporal bone window
- Designed for real-time ultrasound imaging in a clinical environment, including use in the operating room and intensive care unit
- Supports B-mode, color Doppler, and spectral Doppler modes for visualizing cerebral anatomy and blood flow
Claims Under Investigation
Certain capabilities, such as advanced visualization through or around bone for expanded diagnostic purposes, are the subject of ongoing clinical research and are not cleared indications under K230511.
The MAUI K3900 is FDA-cleared for the indications listed in K230511. Some capabilities highlighted are under active clinical study and not claims of cleared indications.

Operational Pathways for Hospitals

Step 1: Request a Demo / Pre-Order
Acertara is managing pre-orders with an exclusive U.S. distributorship for hospitals outside of those MAUI serves directly. Hospitals interested in evaluating the K3900 can contact Acertara’s sales engineering team to arrange demonstrations, site assessments, and procurement discussions.

Step 2: Compatibility & Workflow Planning
Adopting a new imaging modality goes beyond a purchase decision and requires a coordinated workflow project. Clinical engineering teams will need to evaluate:
- Physical footprint and power requirements for the K3900 system
- Connectivity and DICOM integration for image archiving and interoperability
- Sterility protocols, if used intraoperatively
- Training schedules for both clinical and technical staff
Planning at this stage allows the technology to enter service without disrupting patient care.
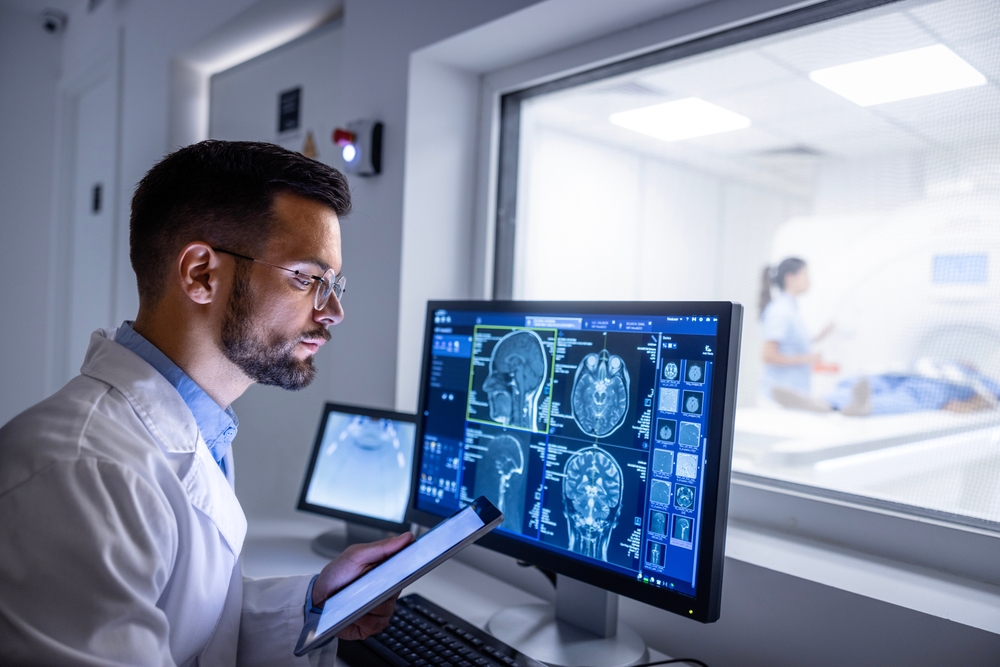
Step 3: QA and Commissioning
Hospitals should treat CET adoption with the same rigor applied to MRI or CT commissioning. Using ATLAS acceptance testing, Acertara can establish a performance baseline at install. This creates a defensible record for quality assurance programs and helps meet accrediting body requirements.

Step 4: Post-Market Support and Service
Because the K3900 uses specialized probes, ongoing performance verification is critical. Acertara’s repair and validation expertise ensures these transducers maintain peak performance over time, reducing downtime and extending device life. Our field data collection also feeds into MAUI’s engineering team, accelerating refinement of future hardware and software updates.
Guesswork isn’t a QA strategy. Partner with Acertara.
FAQs
Is MAUI Imaging a replacement for CT or MRI?
No. CET is a complementary modality designed for situations where ultrasound is indicated but conventional probes are limited by anatomy. It does not replace cross-sectional imaging where that is clinically required.
What is the K-number and cleared indications?
The MAUI K3900 is cleared under FDA 510(k) K230511, which contains the official list of indications for use. Hospitals should review this record to align their use cases with cleared claims.
Does Acertara provide training?
Yes. Training is part of our distributorship role and includes user education, QA integration, and workflow optimization for each clinical environment.
How does CET differ from conventional ultrasound?
CET uses proprietary algorithms and multiple echo paths to create images that may reveal structures that conventional 2D beam-forming misses. This is subject to the FDA-cleared indications and ongoing clinical research.
What does Acertara’s distribution scope mean?
Acertara manages pre-orders and U.S. distribution for hospitals not served directly by MAUI, providing full market coverage while respecting MAUI’s existing direct relationships.
Can it be deployed immediately?
Yes — for cleared indications under K230511. Pre-deployment planning is required to ensure facility readiness, integration, and user training.
Advancing Ultrasound Imaging With CET and Acertara’s Support
The MAUI K3900’s introduction into U.S. hospitals represents a milestone in ultrasound innovation. With its CET technology, MAUI Imaging is addressing clinical scenarios that have challenged conventional imaging for decades.
Acertara’s role is not limited to distribution. We align procurement, training, quality assurance, and service so hospitals can deploy this technology safely, effectively, and in full regulatory compliance.
Hospitals interested in seeing how the K3900 fits into their clinical environment can contact our team to begin the evaluation process.
Reach Us
We want to hear from you about your current probe repair challenges. Drop us an email or call and ask to speak to one of our awesome Customer Service Representatives.
1950 Lefthand Creek Lane, Longmont, CO 80501
+1 303.834.8413


