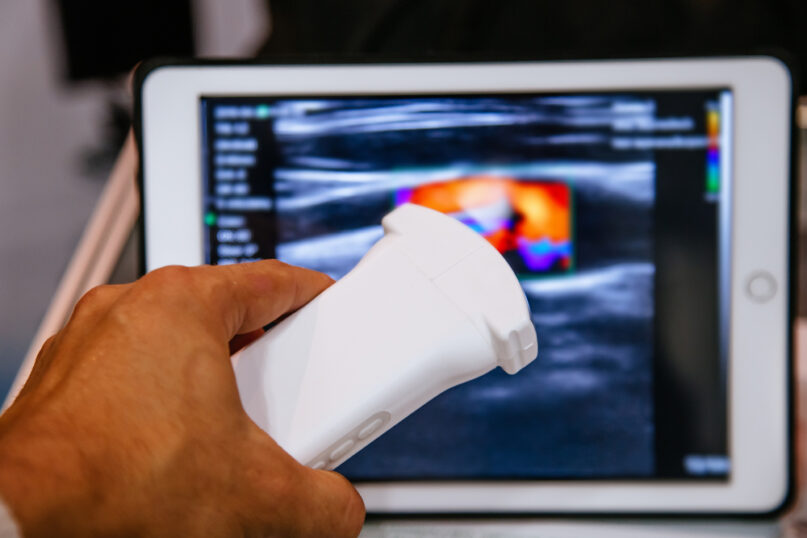
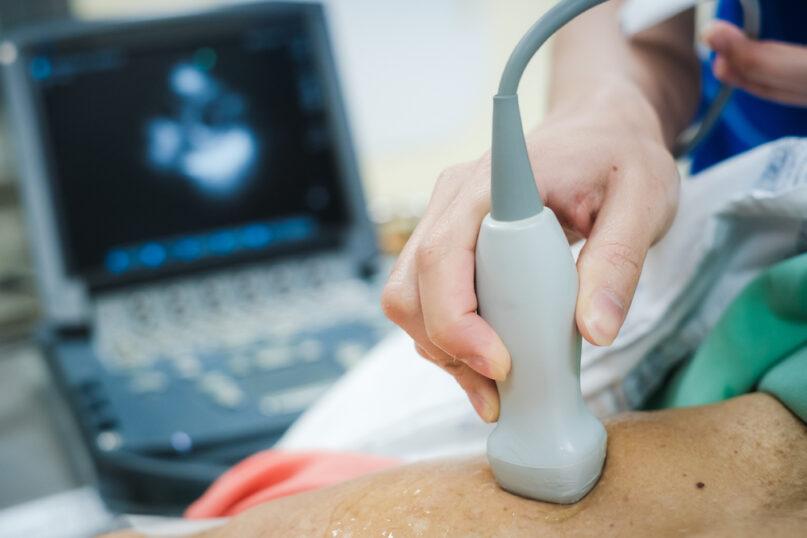
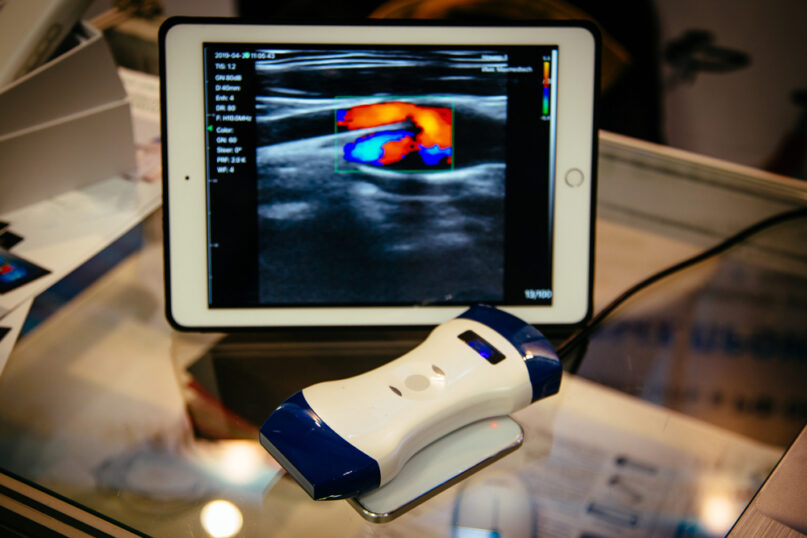
2024’s Compliance Updates for Quality Assurance in Ultrasound
The year 2024 marked significant advancements in regulatory frameworks for medical devices, especially ultrasound systems. The updates are not just about adhering to new rules but are a reminder of the growing need for quality assurance in ultrasound to support patient safety, diagnostic precision, and compliance with FDA requirements. These changes reflect broader trends in … Continue reading